 
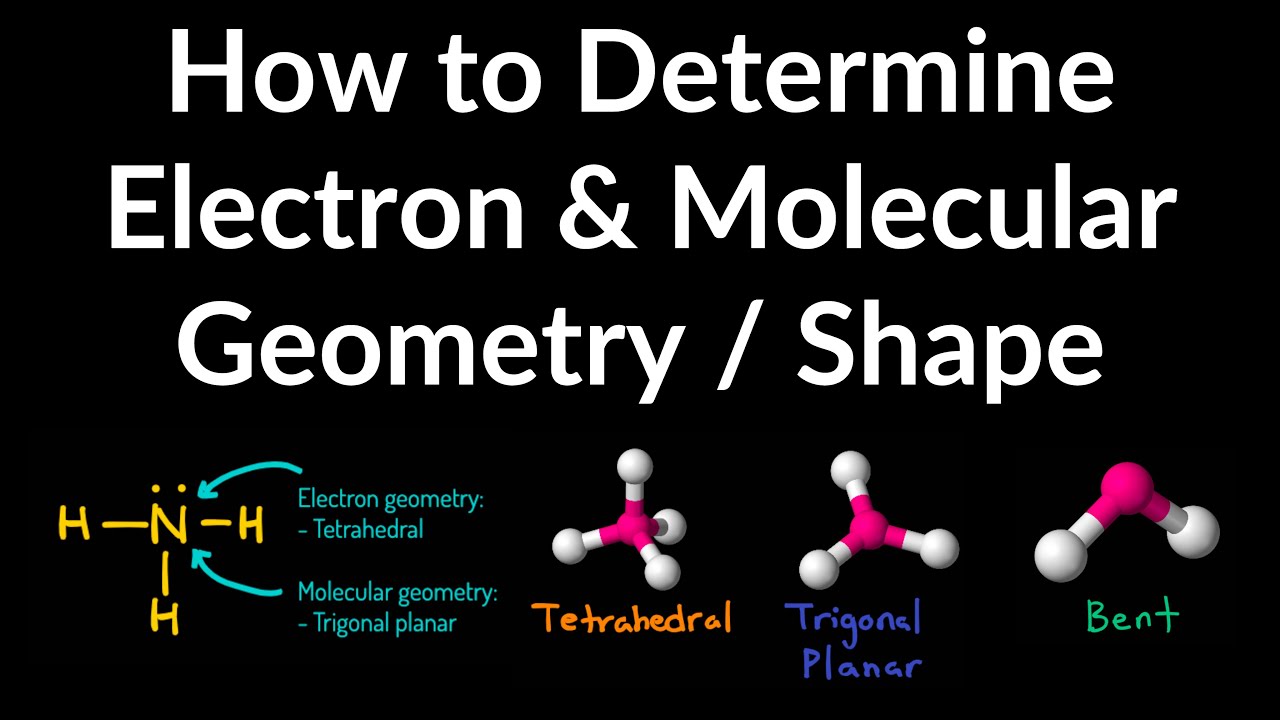
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. The ideal bond angle in AX 2 E type molecule is less than 120º. AX 2 E-type molecules are generally polar. The molecular geometry or shape of AX 2 E-type molecules is bent. The ideal electronic geometry of AX 2 E-type molecules is trigonal planar. By checking the geometry of molecules chart above, we have a tetrahedral shape. In total, 2+1 3 electron density regions are present around the central atom (A). The VSEPR notation for these molecules are AX n. (b) The trigonal pyramidal molecular structure is determined from the electron-pair geometry. 
It gives information about the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. As stated above, molecular geometry and electron-group geometry are the same when there are no lone pairs. 5: (a) The electron-pair geometry for the ammonia molecule is tetrahedral with one lone pair and three single bonds.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
